Almost all the physiological actions of angiotensin II, the effective mediator after renal renin has cleaved the precursor angiotensinogen and the angiotensin-converting enzyme (ACE) of the lung has liberated angiotensin II from angiotensin I, are mediated by by the angiotensin II type 1 receptor. It is a G-protein coupled receptor (GPcR) like many hormone receptors of the rhodopsin family (Omin 106165). Like many GPcR it has been difficult to crystallize to dertermine its threedimensional structure.
Zhang and colleagues from Los Angeles and other places have now used femtosecond chrystallography to reveal the structure with the high-affinity agonist ZD71DD as ligand.
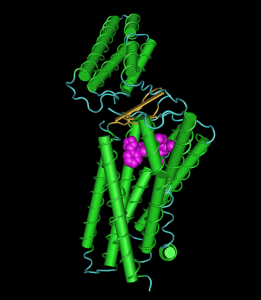
The depicted image is nothing compared to the images in the paper. You can, however, see how the ligand fits in a binding pocket in the transmembrane domain with its numerous helices. The domain on top is a extracellular domain.
The structure should help to resolve questions concerning the regulation of blood pressure, how mutations influence the binding of angiotensin II and may help to develop other drugs.
This is a very nice piece of work. Highly recommended!